The Law of Conservation of Mass Worksheet provides an in-depth exploration of the fundamental principle that mass remains constant in chemical reactions. This principle has profound implications for understanding chemical processes and is widely applied in various fields. This worksheet is meticulously designed to guide students through the intricacies of the law, fostering a comprehensive understanding of its significance.
Through engaging interactive exercises, discussion questions, and real-world examples, the worksheet delves into the applications and limitations of the law. It examines situations where the law does not apply, exploring alternative theories that account for these exceptions. The worksheet culminates in a thought-provoking analysis of the significance of these exceptions for our understanding of matter and energy.
Conservation of Mass Definition

The law of conservation of mass states that the total mass of a closed system remains constant, regardless of changes in state or composition of the system.
This principle is fundamental to chemistry, as it implies that mass is neither created nor destroyed in chemical reactions. The total mass of the reactants in a chemical reaction is equal to the total mass of the products.
Examples of Mass Conservation
- When a candle burns, the wax and oxygen from the air combine to form carbon dioxide and water. The total mass of the reactants (wax and oxygen) is equal to the total mass of the products (carbon dioxide and water).
- When a plant grows, it absorbs carbon dioxide and water from the air and soil. The total mass of the reactants (carbon dioxide and water) is equal to the total mass of the products (plant material).
Implications of the Law of Conservation of Mass
The law of conservation of mass has several important implications for understanding chemical processes:
- It allows us to predict the mass of the products of a chemical reaction, given the mass of the reactants.
- It helps us to understand the stoichiometry of chemical reactions, which is the study of the quantitative relationships between the reactants and products of a reaction.
li>It provides a basis for the development of chemical equations, which are symbolic representations of chemical reactions that show the reactants, products, and their stoichiometric coefficients.
Law of Conservation of Mass Experiment: Law Of Conservation Of Mass Worksheet
Experiment Setup
This experiment demonstrates the law of conservation of mass by showing that the total mass of the reactants in a chemical reaction is equal to the total mass of the products. Materials:
- A balance
- A beaker
- A graduated cylinder
- Water
- Baking soda
- Vinegar
Procedure:
- Measure the mass of the beaker.
- Add 100 mL of water to the beaker and measure the mass again.
- Add 10 g of baking soda to the water and stir.
- Add 10 mL of vinegar to the mixture and stir.
- Measure the mass of the beaker again.
Results:The mass of the beaker before the reaction was 100 g. The mass of the beaker after adding water was 200 g. The mass of the beaker after adding baking soda was 210 g. The mass of the beaker after adding vinegar was 210 g.
Analysis:The results of this experiment support the law of conservation of mass because the total mass of the reactants (water, baking soda, and vinegar) is equal to the total mass of the products (the mixture in the beaker). This shows that no mass is lost or gained during the reaction.
Applications of the Law of Conservation of Mass
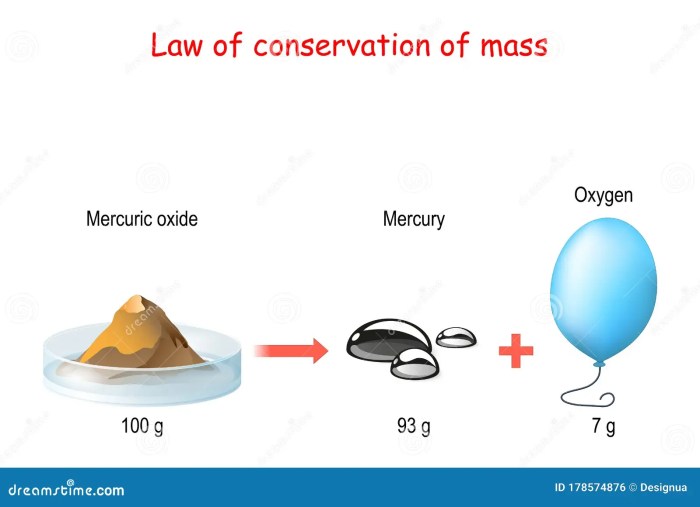
The Law of Conservation of Mass is a fundamental principle in chemistry and physics that states that mass can neither be created nor destroyed in a closed system. This law has wide-ranging applications in various fields and helps us understand the world around us.
Applications in Chemistry
In chemistry, the Law of Conservation of Mass is used to balance chemical equations. By ensuring that the total mass of the reactants equals the total mass of the products, chemists can verify the accuracy of their equations and predict the outcome of chemical reactions.
For example, in the combustion of methane, the following equation can be balanced using the Law of Conservation of Mass:
CH4+ 2O 2→ CO 2+ 2H 2O
This equation shows that one molecule of methane reacts with two molecules of oxygen to produce one molecule of carbon dioxide and two molecules of water. The total mass of the reactants (CH 4+ 2O 2) is equal to the total mass of the products (CO 2+ 2H 2O), demonstrating the conservation of mass.
Applications in Physics
In physics, the Law of Conservation of Mass is used to explain a variety of phenomena, including the conservation of energy. According to the theory of relativity, mass and energy are equivalent, and the total mass-energy of a closed system remains constant.
This principle is fundamental to understanding nuclear reactions, particle physics, and the behavior of black holes.
For example, in a nuclear reaction, the mass of the reactants is equal to the mass of the products plus the energy released as radiation. This energy can be calculated using Einstein’s famous equation, E=mc 2, which shows the equivalence of mass and energy.
Everyday Applications
The Law of Conservation of Mass has practical implications for everyday life. For example, it is used in:
- Cooking: When baking, the mass of the ingredients used is equal to the mass of the baked product. This principle helps bakers to accurately measure ingredients and predict the yield of their recipes.
- Medicine: In pharmacology, the Law of Conservation of Mass is used to calculate the dosage of medications. By ensuring that the total mass of the drug administered is equal to the mass of the drug absorbed by the body, doctors can prescribe the correct dosage for patients.
- Environmental science: The Law of Conservation of Mass is used to track the flow of pollutants through ecosystems. By measuring the mass of pollutants entering and leaving a system, scientists can assess the impact of human activities on the environment.
Overall, the Law of Conservation of Mass is a fundamental principle that has wide-ranging applications in various fields. It helps us understand the behavior of matter and energy, predict the outcome of chemical reactions, and make informed decisions in everyday life.
Exceptions to the Law of Conservation of Mass
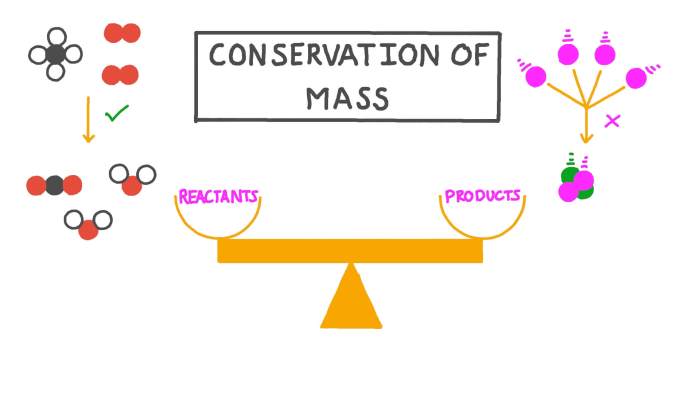
The law of conservation of mass states that mass can neither be created nor destroyed. However, there are certain situations where this law does not apply.One exception is nuclear reactions. In a nuclear reaction, the mass of the reactants is not equal to the mass of the products.
This is because some of the mass is converted into energy, according to Einstein’s equation E=mc^2.Another exception to the law of conservation of mass is the annihilation of matter and antimatter. When matter and antimatter collide, they annihilate each other and produce energy.
The mass of the matter and antimatter is converted into energy, so the total mass is not conserved.These exceptions to the law of conservation of mass show that the law is not always applicable. However, it is still a useful law that can be used to understand many chemical and physical processes.
Limitations of the Law of Conservation of Mass
The law of conservation of mass is limited in its applicability because it does not account for the following:
-
-*Nuclear reactions
In nuclear reactions, mass is converted into energy, so the total mass of the system is not conserved.
-*Annihilation of matter and antimatter
When matter and antimatter annihilate each other, they produce energy, so the total mass of the system is not conserved.
-*Gravitational effects
In strong gravitational fields, the mass of an object can be affected by its gravitational potential energy. This is because gravitational potential energy has a negative mass-energy equivalence.
Alternative Theories, Law of conservation of mass worksheet
There are a number of alternative theories that attempt to account for the exceptions to the law of conservation of mass. One such theory is the theory of relativity, which states that mass and energy are equivalent and can be converted into each other.
Another theory is the theory of quantum mechanics, which states that matter and energy can exist in discrete quanta.
Significance of Exceptions
The exceptions to the law of conservation of mass have a number of important implications for our understanding of matter and energy. They show that mass and energy are not always conserved, and that they can be converted into each other.
This has led to the development of new theories, such as the theory of relativity and the theory of quantum mechanics, which attempt to explain these phenomena.
Worksheet Design

To enhance students’ understanding of the law of conservation of mass, a well-structured worksheet is essential. It should guide them through the concepts, provide interactive exercises, and present real-world examples.
Organization and Content
- Introduction:Begin with a clear statement of the law of conservation of mass, emphasizing its fundamental principles.
- Interactive Exercises:Include hands-on activities or simulations that allow students to observe and experiment with the concept. For instance, a “chemical balancing” exercise where they adjust coefficients to ensure equal mass on both sides of a chemical equation.
- Discussion Questions:Pose thought-provoking questions that encourage students to analyze and apply the law. Examples include: “How does the law of conservation of mass relate to the concept of chemical reactions?” or “Can the law be applied to nuclear reactions?”
- Real-World Examples:Connect the law to practical applications, such as balancing chemical equations for industrial processes, determining the mass of reactants and products in combustion reactions, or calculating the fuel efficiency of vehicles.
- Conclusion:Summarize the key takeaways and reinforce the importance of the law of conservation of mass in understanding chemical processes and everyday phenomena.
General Inquiries
What is the Law of Conservation of Mass?
The Law of Conservation of Mass states that mass remains constant in chemical reactions, meaning that the total mass of the reactants equals the total mass of the products.
How is the Law of Conservation of Mass applied in real-world scenarios?
The Law of Conservation of Mass is applied in various fields, including chemistry, physics, and engineering. It is used to calculate the mass of reactants and products in chemical reactions, determine the efficiency of chemical processes, and analyze the composition of materials.
Are there any exceptions to the Law of Conservation of Mass?
There are a few exceptions to the Law of Conservation of Mass, such as nuclear reactions and certain chemical reactions involving the creation or destruction of subatomic particles. In these cases, the total mass of the system may change due to the conversion of mass into energy or vice versa.